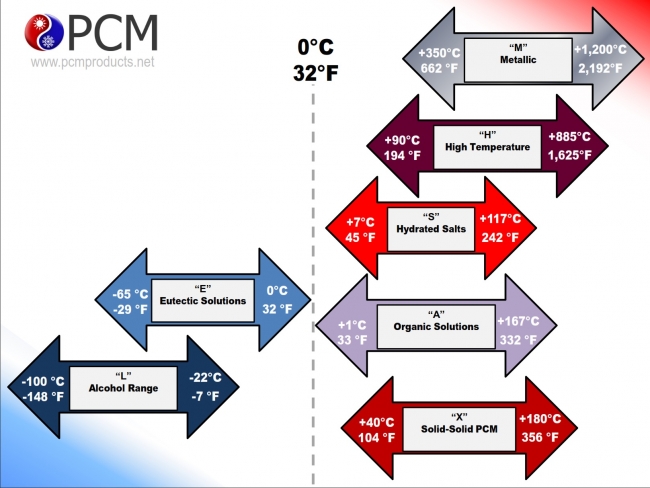
Our PlusICE range of PCM solutions and associated products cover a wide range of applications between -100°C (-148°F) and +885°C (+1,625°F) and are available either as the standard PCM solution, or in a variety of formats and encapsulated versions.
Find out more
Our PlusICE range of products including FlatICE, BallICE, TubeICE, Eutectic Plates and Pouches enables us to develop and apply both organic and hydrated salt based PCMs for a variety of applications around the world.
Find out moreCONSULTANCY

PCM Products Ltd. is actively involved on R&D and new product development projects with many Industrial and Academic institutions around the World.
Find out morePhase Change Materials: Thermal Management Solutions
An introduction to Phase Change Materials
Phase Change Materials (PCMs) are ideal products for thermal management solutions. This is because they store and release thermal energy during the process of melting & freezing (changing from one phase to another). When such a material freezes, it releases large amounts of energy in the form of latent heat of fusion, or energy of crystallisation. Conversely, when the material is melted, an equal amount of energy is absorbed from the immediate environment as it changes from solid to liquid.
This property of PCMs can be used in a number of ways, such as thermal energy storage whereby heat or coolness can be stored from one process or period in time, and used at a later date or different location. PCMs are also very useful in providing thermal barriers or insulation, for example in temperature controlled transport.
The simplest, cheapest, and most effective phase change material is water/ice. Unfortunately, the freezing temperature of water is fixed at 0°C (32°F), which makes it unsuitable for the majority of energy storage applications. Therefore a number of different materials have been identified and developed to offer products that freeze and melt like water/ice, but at temperatures from the cryogenic range to several hundred degrees centigrade.
Our PCM range can broadly be arranged into three categories: eutectics, salt hydrates, and organic materials.
- Eutectics tend to be solutions of salts in water that have a phase change temperature below 0°C (32°F).
- Salt hydrates are specific salts that are able to incorporate water of crystallisation during their freezing process and tend to change phase above 0°C (32°F).
-
Organic materials used as PCMs tend to be polymers with long chain molecules composed primarily of carbon and hydrogen. They tend to exhibit high orders of crystallinity when freezing and mostly change phase above 0°C (32°F). Examples of materials used as positive temperature organic PCMs include waxes, oils, fatty acids and polyglycols.
-
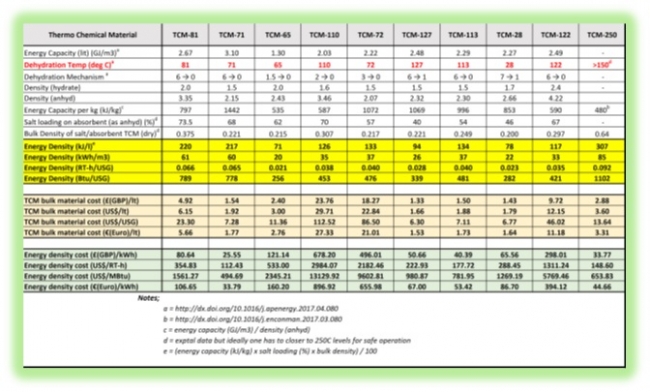
Thermo Chemical Material - TCM energy storage may yield a reasonable heat storage capacity without producing any thermal losses during the storage period. The working pairs of various salt options incorporated in high porous structured carrier materials whereby utilising a reversible chemical reaction and takes the advantages of strong chemical bonds to store energy as chemical potential. Compared to sensible heat storage and latent heat storage, this theoretically offers higher energy density with minimum energy loss during long-term storage due to the temperature-independent means of storage.Following extensive development programme over the last 10 years it is established that the most critical aspect of the Thermo Chemical Material (TCM) based Thermal Energy Storage (TES) is the regeneration temperature of the TCM. Hence, the following range of TCM materials are designated based on the regeneration point.
Product Development
Over the last two decades we have succesfully developed a wide variety of applications for our customers around the World and the relevant details can be see in the following examples and for further details just click the relevant pictures.